If you are interested in learning about cannabis as medicine, you are bound to come across specific terms, jargon, or names that are important to learn about but which maybe new or even strange to you. This is especially true if you want to consider the currently available scientific literature to determine if cannabis is posited to work for a particular condition or not. One of those terms is cannabinoid. You’ll find it as a stand-alone term or expressed with various prefixes to indicate significant differences: endo-, exo-, phyto- pharmaceutical- and synthetic-cannabinoids. In this article we will explore each of these terms, what they mean and how they differ and why each of them is important especially when it comes to generating medical and therapeutic effects.
What is a Cannabinoid?
In general, the term cannabinoid is derived combining two words i.e., cannabis and the suffix -oid taken from the ancient Greek –oeides meaning likeness to indicate alike. As such, the meaning is straightforward: “like or similar to cannabis.” Cannabinoids are further defined by where or how they are sourced. If they are sourced or made by the body itself, they are termed endocannabinoids. The prefix endo- is taken from the Greek éndon meaning inner or within. The most studied and arguably significant endocannabinoids are anandamide (AEA) and 2-arachidonoyl glycerol (2-AG). Cannabinoids sourced outside the body are referred to as exocannabinoids. The Greek prefix exō- meaning external or outside. These include phytocannabinoids (phyto- from the Greek word phyton meaning plant), those made by the pharmaceutical industry (prescription drugs), and those that are produced artificially (synthetic cannabinoids). Additionally, you may also have come across the term cannabinomimetic, which indicate compounds that mimic the effects of cannabinoids. A couple of commonly known examples of the latter would include Echinacea or Copaiba oil for example. As an easy to remember reference, we have included this table of definitions:
Cannabinoid Groups
| Endocannabinoids (Produced within the body) | Exocannabinoids (Produced outside the body) |
|---|---|
| Anandamide | Phytocannabinoids (cannabis) |
| 2-Arachidonoyl Glycerol | Synthetic cannabinoids (man-made) |
| Pharmaceuticalcannabinoids (drugs) |
General Differences Between Groups of Cannabinoids
Here are few general differences or characteristics between the broad types or groups of cannabinoids that help to better understand their importance recreationally and of course in the clinical setting.
- Endocannabinoids are produced naturally by our bodies. You can consider endocannabinoids essential compounds that help to maintain overall balance and resilience. Lower endocannabinoid levels are associated with ill-effects.
- Plant-based cannabinoids have been part and parcel of recreational and medicinal purposes for thousands of years. Phytocannabinoids in all their forms have become an essential tool in the clinical setting.
- Man-made versions play substantial roles in research where they are used to help us understand how to harness and direct their impact on the human body and mind. However, man-made cannabinoids are also readily available for purchase on the internet and have found their way into several recreational products many of which have been implicated in severe injury and even death [see warning about synthetic cannabinoids issued by the Centers for Disease Control and Prevention (CDC)at the end of article].
- Pharmaceutical cannabinoids can either be plant-based or entirely synthetic. They are available by prescription wherever legal and have been used extensively and effectively in treating diverse patient populations suffering from chronic illness typically poorly served by other pharmaceuticals.
Specific Differences Between Groups of Cannabinoids
The differences between groups of cannabinoids are defined by:
- Where they are produced (which as you have seen also determines their specific name)
- The impact they have physiologically and psychologically
- Their therapeutic potential
- Their potential to produce adverse effects
- Where we are likely to encounter them
- Their unique pharmacodynamics (what the drug does to the body) and pharmacokinetics (how body processes the drug) measured by their affinity for corresponding receptor sites (Ki).
The latter measure (Ki) has significance because some cannabinoids bind to certain receptor sites while not with others. Furthermore, some cannabinoid binds to certain receptor sites with different intensities. For instance, numerous synthetic cannabinoids used in recreational products have a high impact at CB1 receptors and as such are responsible for numerous serious effects including organ failure and death. In direct contrast natural cannabinoids have either a very mild or moderate impact at CB1. Scientifically, these impact variations of different cannabinoids are measured in terms of affinity or efficacy. Affinity is a measure that describes if and how tightly a specific cannabinoid binds with a specific receptor. Efficacy on the other hand is a measure that is commonly used to describe the ability of a cannabinoid to activate a receptor once binding has occurred and with it the effects that is subsequently being generated such as producing an anti-inflammatory response for example. How is affinity or efficacy measured? Simply put, there are four categories of measures ranging from strong, moderate, weak, to very weak.
Cannabinoids—An Overview
| Name | Endo- | Phyto- | Pharma- | Synthetic- |
| Examples | Anandamide | THC | Sativex | HU-210 |
| Means of production | Endogenous (naturally occurring) | Plant-based | Man-made | Man-made |
| Plays a role in | Self-regulation | Clinical setting | Clinical setting | Research, (some recreational products) |
| Impact on | Mind and body | Mind and body | Mind and body | Mind and body |
| Therapeutic potential | Moderate-high | Moderate- high | Moderate- high | TBD |
| Affinities/Efficacy | Weak | Moderate | Moderate | Strong |
| Ki | 239.2nM at CB1 439.5nM at CB21 | 25nM at CB1 35nM at CB22 | THC moderate,3 CBD very weak4 | ~0.25nM at CB1 ~0.40nM at CB25 |
| Affinity/Efficacy associated severe adverse effects potential | N/A | Low-moderate risk (in some THC-sensitive people) | Low risk, moderate risk in some THC-sensitive people | High risk |
| Access | Mind-body training | Dispensaries, home grown | Prescription drugs (Rx) | Researchers, internet |
As such, the more we know and understand the different effects that each grouping of cannabinoids can produce the more we are able to create the precise effects we deem therapeutic, recreationally desirable and avoid those we consider adverse. The more we learn to harness cannabinoids the more we can generate a predictability of effects. Due to the abundance of individual cannabinoids to cover all of them would be beyond the scope of this article. However, to highlight the uniqueness of each group we chose one of each to take a closer look. Here we review the endocannabinoid anandamide (AEA), the phytocannabinoid delta-9-tetrahydrocannabinol (THC), the man-made HU-210, and the pharmaceutical cannabinoid Sativex.
Endocannabinoids
Anandamide
The first endocannabinoid was isolated and revealed to the scientific community in 1992 by Rafael Mechoulam at the International Cannabis Research Society Symposium. The team decided to name the compound “anandamide” by combining the Sanskrit word for “bliss,” ananda, and the scientific word amide, describing an organic chemical compound. You may also find it under its nickname “the bliss molecule.”
Our bodies produce and degrade (break down) anandamide on an as-needed basis to maintain or establish balance (homeostasis). To date, nearly 200 studies have examined the properties of AEA, with more than a dozen of them being clinical trials conducted on humans.
The scientific literature points to a handful of receptor sites (CB1, CB2, PPAR-α and -y, TRPV1, and 5-HT/serotonin receptors) through which AEA is posited to produce its effects on body, mind, and emotions. Anandamide is a weak agonist at both CB1 and CB2 receptor sites (at CB1 ~240nM and at CB2 ~440nM). But this does not mean that its therapeutic actions are weak. Its effects play a therapeutic role in mitigating pain, improving mood and cognition, and reducing inflammation and oxidative stress for instance.
In the presence of the corticosteroids (steroidal hormones involved in several functions such as stress responses), the body increases its production of anandamide, which may account for the way in which anandamide boosts the antidepressant effects of exercise.
Additional health benefits of anandamide include a potential for reducing depression, fear, and anxiety as well as contributing to social play behavior, making it relevant in the treatment of psychiatric disorders characterized by impairments in social behavior such as autism or schizophrenia. Anandamide enhances the experience of pleasure derived from food. Several pre-clinical trials have shown that AEA can destroy numerous types of cancers in the laboratory including the potential for preventing metastasis.
AEA Has Shown Proven Effects in Clinical Human Trials for These Conditions
| Epilepsy | Post-Traumatic Stress Disorder |
| Hypertension and Hypertensive Diseases | Psychosis |
| Libido | Schizophrenia |
| Motion Sickness | Stress and Life Management Difficulty |
| Multiple Sclerosis | Vomiting in Pregnancy |
Exocannabinoids
Phytocannabinoids Cannabinoids (Plant-based)
Plant-based cannabinoids are almost exclusively associated with cannabis.1 Currently, research has chronicled the existence of more than 150 cannabis-based cannabinoids that are typically divided into 10 main and distinct sub-groups:
Phytocannabinoid Groups (Alphabetically)
| Cannabichromene (CBC) | Cannabinol (CBN) |
| Cannabidiol (CBD) | Cannabinodiols (CBND) |
| Cannabielsoin (CBE) | Cannabitriol (CBT) |
| Cannabigerol (CBG) | Δ8-tetrahydrocannabinol (Δ8-THC) |
| Cannabicyclol (CBL) | Δ9-tetrahydrocannabinol (Δ9-THC) |
Of all the known phytocannabinoids to-date only four have been tested in the clinical setting. Of these, Δ-9- Tetrahydrocannabinol (Δ -9- THC) and Cannabidiol (CBD) are the most studied cannabinoids. For instance, by December 2021, more than 192 clinical trials (i.e., conducted on humans) directly examined the effects of THC. In fact, both THC and CBD have demonstrated therapeutic effects potential in the treatment of numerous, especially chronic patient population (see list below). Two of the new commers to the clinical setting are Tetrahydrocannabivarin (THCV) explored as potential treatment options for patients challenged by metabolic syndrome, obesity, and diabetes, and Δ-8- Tetrahydrocannabinol (Δ-8-THC) in the treatment context of nausea and vomiting for example.
Here we briefly review Δ-9-THC.
Δ-9- Tetrahydrocannabinol (THC)
Delta-9-Tetrahydrocannabinol is the primary mind-altering constituent of cannabis, responsible for generating complex changes that occur physically as well as mentally and emotionally. The chemical structure of THC was discovered in 1964 by two Israeli scientists, Raphael Mechoulam and Yechiel Gaoni. THC is a multi-target cannabinoid that binds to receptor sites in all organ systems and as such weaves a complex web of specific effects that are relevant to a great number of different patient populations, especially those that share chronic inflammation, oxidative stress, and chronic pain.
Researchers have identified more than 50 pharmacological actions that affect body, mind, and emotions that are realized via the modulation of a diverse group of THC-sensitive receptor sites that include, but are not limited to: CB1, CB2, GPR18, GPR 55; opioid receptor sites (via allosteric modulation); various transient receptor potential channels (e.g., TRPA1, TRPV2, TRPM8; numerous neurotransmitter receptor sites activation via (+ or -) allosteric mechanisms (e.g. serotonin, glutamate, epinephrine), as well as peroxisome proliferator-activated receptor (PPAR-gamma).
More practically speaking, THC-abundant cannabis plants or products (chemotype I) have proven effects that dependent on a patient’s sensitivity to THC and can range between a powerful therapeutic or an adverse effect. Therapeutic examples include bronchodilation (CB1 inhibits cholinergic contraction of bronchi), appetite stimulation, down-regulation of the body’s own immune responses, and potent analgesic properties in cases of central pains. As such they have been employed therapeutically in patients with asthma, HIV/AIDS-related anorexia, rheumatoid arthritis, or spinal cord injuries (for more examples see list below). On the other hand, the scientific literature clearly suggests caution for patients with pancreatitis, individuals with vulnerabilities to developing psychotic events, adolescents, or during pregnancy for instance. Due to space here is a listing that covers diseases from A-C only.
THC Has Shown Proven Effects in Clinical Human Trials for These Conditions (A-C only)
Synthetic Cannabinoids
HU-210
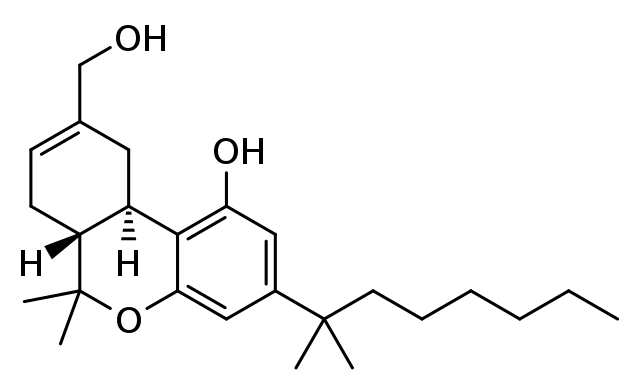
More than a hundred of synthetic cannabinoids have been produced, characterized, and studied to better understand the complex workings of the ECS in the context of the healing arts. However, many of these compounds can be purchased legally on the internet; and sadly, many end up being sold in unregulated and dangerous ways to customers unfamiliar with the very real and potential dangers to their health and well-being. Those with the biggest practical implications in medicine are synthetic cannabinoids that show up in “street drugs” such as “Spice,” K2, or synthetic marijuana, for example.
Here we review one of those synthetic cannabinoids: HU-210. HU stand for Hebrew University and 210 is a designation to differentiate between various HU-based compounds. HU-210 was created in 1988. You may have come across news reports that clearly implicate synthetic cannabinoids in overdoses so severe that they caused organ failure and death. The reasons why many synthetic cannabinoids are dangerous when compared to their naturally occurring counterparts has to do with the concept of binding affinity. Both naturally occurring and synthetic cannabinoids bind with endocannabinoid receptor sites such as CB1. However, the latter bind with an affinity that is significantly higher, with direct and potentially devastating consequences of resulting effect. When comparing THC with HU-210 you will notice that THC binds with moderate affinity, which is high enough (no pun intended) to induce changes in cognition (even hallucinations) but not high enough to kill. In direct contrast, the affinity of HU-210 (a purported ingredient in “Spice”) is about one hundred times higher at the same receptor site, thus it can cause increasingly potent effects in the central nervous system and any other bodily system that contains CB1 sites that can include organ failure and death.
At the writing of this article 49 pre-clinical trials have been published directly examining the effects of various HU-related compounds. Clinical trials remain wanting (as previously mentioned but worth repeating [see warning about synthetic cannabinoids issued by the Centers for Disease Control and Prevention (CDC)at the end of article].
Pharmaceutical Cannabinoids
Nabiximols (THC:CBD)
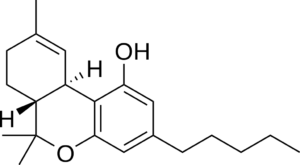
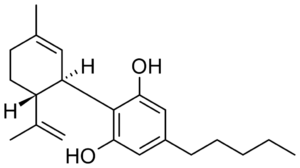
Pharmacological products containing cannabinoids currently correspond to or are analogues of the three cannabis chemotypes I, II, and III. A chemotype I contain predominantly THC, a chemotype II contains relatively equal amount of THC and CBD (roughly in ratios between 4:1 and 1:4 respectively), and a chemotype III contains primarily CBD with only small or trace amounts of THC. Since we covered THC and CBD in the previous sections here, we will review the cannabis chemotype II variety.
They represent a class of drugs that are produced and standardized from either man-made or plant-based materials. For instance, Nabiximols, trade name Sativex (brand names Almirall, Almirall Hermal, Mevatyl) is a plant-based extract from cannabis. Sativex is a standardized pharmaceutical product that delivers relative equal amount of THC and CBD in a sublingual spray (each actuation delivers ~2.7mg THC and 2.5mg CBD). Sativex is legal in twenty-five countries around the world.
To-date 33 clinical trials have directly examined the effects of Nabiximols in the clinical setting.
Nabiximols (or analogues) Have Shown Proven Effects in Clinical Human Trials for These Conditions
To conclude, the differences between the various groups of cannabinoids can be summarized by one common and practically relevant factor. The impact they have on the human body. Endocannabinoids are essential to inducing homeostasis (balance). And there are several steps we can take to support a healthy ECS such as getting enough sleep, eating healthy food, exercise, reducing stress naturally by practicing a mindfulness technique of your choice.
Phytocannabinoids, whether in plant or pharmaceutical form, have become an essential stress relieving agent around the world and a much-appreciated novel remedy in the clinical setting. In the academic or research setting man-made cannabinoids continue to provide researchers with the ability to better understand the detailed underpinning of the ECS. When it comes to synthetic cannabinoids in any other context consider the advice of the CDC (see the following link or the text in the grey box below): https://www.cdc.gov/nceh/hsb/chemicals/sc/default.html
Warning! The Illinois Department of Public Health (IDPH) has received reports of multiple cases, including some deaths, of severe bleeding among people who have used contaminated synthetic cannabinoids. Other states also have reported similar cases. If you have purchased any of this product, which go by names such as (K2, spice, and synthetic marijuana) do not use it. Throw it out. If you have used any of these products and start experiencing severe, unexplained bleeding or bruising, please have someone take you to the hospital immediately or call 911. Do not walk or drive yourself. Tell your health care or emergency care providers that you have used synthetic cannabinoids. Even if you don’t have symptoms, tell your medical care provider about any synthetic cannabinoid use if you are having a medical procedure. Learn more about the IDPH cases here. See CDC’s two recent Clinical Action alerts here and here
Endnotes to Table: Cannabinoid—An Overview
1. McPartland JM, Glass M, Pertwee RG. Meta-analysis of cannabinoid ligand binding affinity and receptor distribution: interspecies differences. British Journal of Pharmacology. 2007;152(5):583-593.
2. Ibid.
3. Ibid.
4. Pertwee R. G. (2008). The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: delta9-tetrahydrocannabinol, cannabidiol and delta9-tetrahydrocannabivarin. British journal of pharmacology, 153(2), 199–215.
5. McPartland JM, Glass M, Pertwee RG. Meta-analysis of cannabinoid ligand binding affinity and receptor distribution: interspecies differences. British Journal of Pharmacology. 2007;152(5):583-593.