While our bodies can’t live forever, we can certainly work to push the boundaries of a commonly expected physical life span from seventy or seventy-five to exceed a century. And I’m not just talking about living longer while being frail, fragile, or feeble, but being healthy, full of life, having resilience, the richness of experience, wisdom, and a high energy level. With a better understanding of the aging process and the rapidly growing scientific knowledge on reversing it, we now have options our parents could have only dreamed about. And, as you will see, inducing carefully crafted changes in the endocannabinoid system (ECS), with or without using cannabis-based therapeutics, may play a significant role in these novel life-extension modalities. Before we dive and explore what these new pathways to longevity might be, it may be helpful to take a brief look at both the science of gerontology–the study of aging, and a relative newcomer to the healing arts–the cannabinoid health sciences.
Gerontology–The Science of Aging
With all our technological advances, it may be a surprise that scientists still need to understand the precise mechanisms of the aging process. Various theories have been proposed to explain the loss of cellular integrity we recognize as aging (see signs and symptoms below). Such approaches primarily focus on inadequate immune responses after exposure to parasites, fungi, bacteria, viruses, or other pathogens; dysfunctional autophagy (allowing for the accumulation of older or damaged cells); mitochondrial dysfunction [i.e., the energy-producing organelle that drives cellular energy levels and resulting (in)activity]; chronic or acute mental-emotional stress, physiological stressors such as the accumulation of toxins, the long-term effect of ionizing radiation, damage from exposure to free radicals (oxidative stressors); changes in hormone profiles; shortening of the end strings of DNA (i.e., telomeres, which occurs with each cell division and in the presence of stressors); and changes within the ECS1.
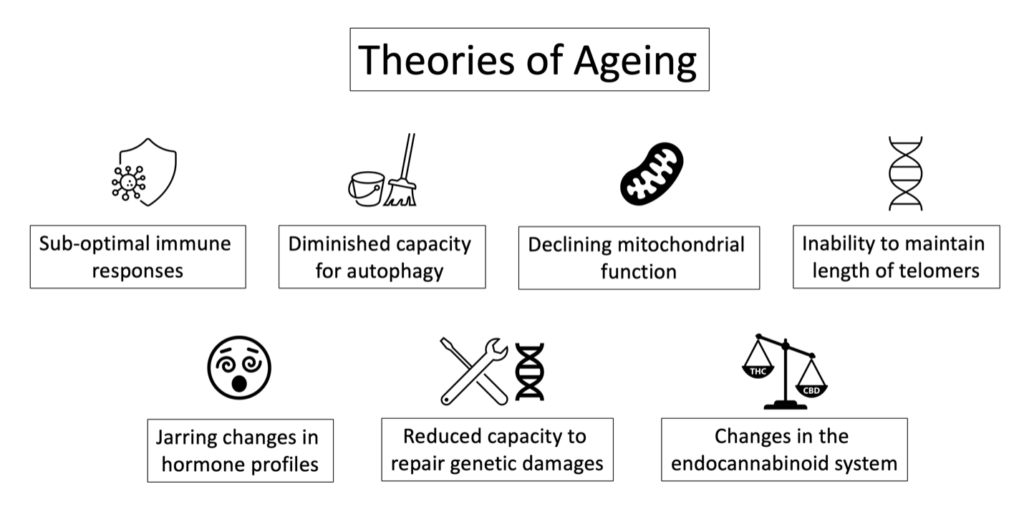
With these changes, we begin to experience suboptimal function at the individual cell level and the whole organism. In the case of the individual cell, the mitochondria start to show degrees of dysfunction, resulting in a reduction in cellular energy production, an increase in mitochondrial waste (i.e., mitochondrial toxicity), loss of mitochondrial homeostasis, homeostatic dysfunctions, reduced capacity to protect against oxidative stress and oxidative damage for instance. If not corrected, this dysfunction can become a driver making us vulnerable to developing a great variety of chronic conditions such as neurological disease, immune disorders, cardio-vascular illness, and aging. In addition, the whole body begins to manifest aging-related signs and symptoms.
Conversely, looking at life extension or longevity, several methods or processes have been posited to extend life at the level of the individual cell and for the whole organism. Here in this article, we center our focus on ECS-based effects associated with the prevention and reversal of cognitive decline (e.g., dementia), facilitation of molecular clearance (i.e., optimization of autophagy, e.g., Alzheimer’s disease), homeostatic defense activities, protection against neurodegeneration and neuro-inflammation (incl. neurogenesis), mitigation of oxidative stress, SIRT activation (a signaling protein with high potential for life extension), caloric restrictions (metabolism of fat cells is modulated by the ECS), mind-body approaches (the ECS modulates stress), and other therapeutic effects induced via the endocannabinoidome (the larger internal environment that interacts with the ECS).
The Cannabinoid Health Sciences (Endocannabinology)–
The Study of the Endocannabinoid System and Cannabinoid-based Therapeutics
The cannabinoid health sciences focus on studying the endocannabinoid system (ECS). The fact is that all mammals—and that includes us humans—have a naturally occurring ECS.
The ECS is composed of receiving elements (cellular receptor sites, i.e., CB1, CB2), their corresponding signaling components (i.e., endocannabinoids anandamide and 2-AG), and their essential building and degrading constituents (e.g., enzymes FAAH, MAGL). And while the ECS has been with us since time immemorial playing an indispensable role in maintaining our health and well-being, modern medicine discovered its existence and, perhaps more importantly, its vast therapeutic potential only in the late 1980s. The first evidence of the presence of a mammalian receptor that binds with cannabinoids and cannabis-based constituents was discovered in the brains of rats (1988) when government researchers from the National Institutes of Mental Health (Bethesda) characterized a cannabinoid receptor that a few years later would be classified as cannabinoid receptor 1 (CB1).1 A few years later, researchers from Cambridge University discovered the second later to be identified as CB2.2
Since then, the number of annual studies published in the attempt to understand and utilize the ECS in the potential treatment of more than 250 conditions has grown considerably. By 2022 an average of about ten new studies have been published daily, signaling the excitement in the medical research community eager to make a difference in the lives of many chronic patient populations, including those affected by signs and symptoms of aging.
The prefix endo- is taken from the Greek éndon, meaning inner or within. The middle section of the word, -cannabis-, is brought to reference cannabis. The suffix -oid is taken from the ancient Greek –oeides meaning likeness, to indicate alike. As such, if we were to line up these words and their respective meanings, we would get “the system that is like cannabis within, or endocannabinoid system.”
Signs and Symptoms of Aging
Physical signs and symptoms: Wrinkles, “age spots,” greying hair, loss of muscle mass, loss of height, weight gain (associated with metabolic syndrome, insulin resistance, diabetes), loss of visual focus, loss of energy (i.e., at the level of the cell and the whole body), longer recovery times, male/female menopause, epigenetic changes, mutation, telomere shortening, loss of genetic stability and DNA repair capacities, loss of optimal organ/system functions, dysfunctional autophagy, mitochondrial dysfunction, dysfunction of the ECS, increased vulnerability to developing chronic degenerative illness associated with aging such as cancer, cardiovascular diseases, osteoporosis, and obesity for example.
Mental-emotional signs and symptoms of sub-optimal aging: may include cognitive decline or dysfunction (e.g., on a spectrum between forgetfulness–dementia), social isolation (e.g., loneliness, hopelessness), mood disorders (e.g., depression, anxiety, rapid fluctuations in mood), ruminating (esp. at night), lack of mental-emotional energy and focus, loss of social skills and functions.
The Endocannabinoid System and Aging in General
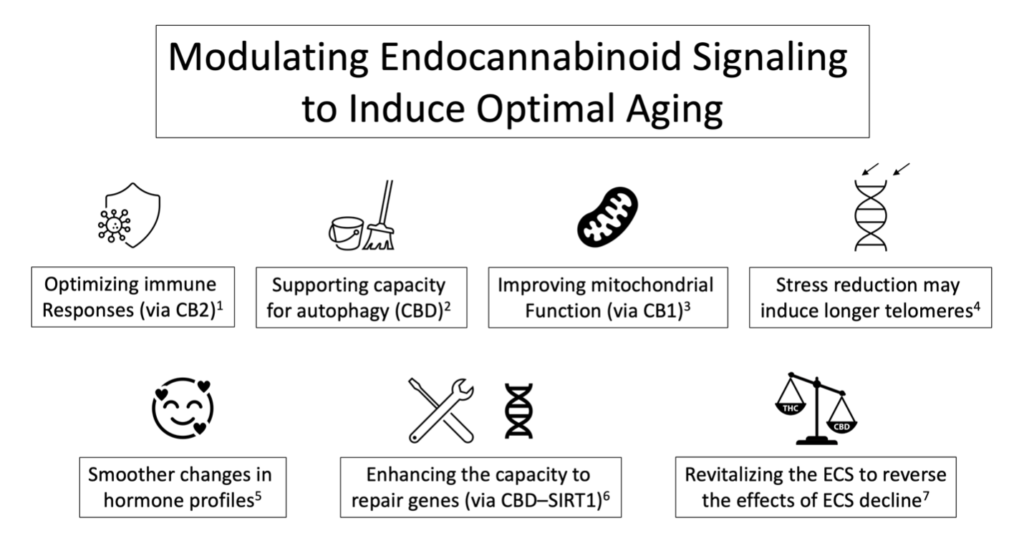
Over the past three decades, the therapeutic potential of modulating the ECS for the treatment of aging and age-related human pathologies has gained momentum and significant interest, especially in the areas of cognitive decline (e.g., dementia, Alzheimer’s disease), facilitation of molecular clearance, homeostatic defense activities, neurodegeneration and inflammation, neurogenesis, and oxidative stress. Optimal function or dysfunction of the ECS, such as modulation of the classical endocannabinoid receptor sites CB1 and CB2, or other cannabinoid-sensitive receptor sites such as TRPV-1, may play a significant role in how we age.
More specifically, the capacity of the body to produce endocannabinoids, metabolize them, and actual endocannabinoid receptor expression; with it, optimal ECS-based signaling can change significantly as we age. You may ask why it is that important. For example, a reduction in endocannabinoid receptor sites has been associated with accelerated aging. Therefore, the targeted use of cannabis-based therapeutics to support optimal ECS function and optimal homeostatic responses may mitigate several underlying pathologies associated with the aging process. Consider these recent discoveries highlighting several emerging insights with potential clinical applications that allow us to make informed decisions about the use of cannabis-based therapeutics to shift the typically pathological aging process to one of optimal aging:

- Expression of cannabinoid receptor sites and cannabinoid signaling changes as we age.1 More specifically, CB1 expression decreases by ~50% over a human life span,2 impairs neuronal network integrity and cognitive functions.3
- Rodents bred without CB1 receptor sites show signs of accelerated aging in the brain and skin.4
- The ECS supports neuroprotection, is neuro-supportive, reduces oxidative stress, stabilizes glial activity, mitigates glutamate excitotoxicity, aids in metabolic homeostasis, and autophagy, all of which are associated with aging.5
- A decline of ECS-based signaling on GABAergic neurons may lead to accelerated brain aging.6
- Deletion of CB2 leads to severe osteoporosis in the test animals, indicating a significant role of CB2 in maintaining healthy bone structures in aging mammals.7
- Enhancing CB18 and CB29 signaling enhances neurogenesis, reduces neuronal decline, and reduces the ill effects of pathological aging upon neuroinflammation.10
- Cannabinoids and melatonin induce therapeutic effects fundamentally related to their anti-inflammatory and antioxidative actions at the mitochondrial level.11
- CB1 activity in the mitochondria declines with age and is associated with signs of aging, such as impaired memory. ECS-based activity may counteract the progression of brain aging.12
- CBD exposure to zebrafish in utero increased longevity by about 20% (in females), reduced inflammation, and decreased sperm production.13
- Only lower doses of THC increased longevity, significantly reduced weight and inflammation, and reduced several senescence markers in male zebrafish.14
- Cannabis-based therapeutics may effectively treat nighttime agitation,15 disinhibition, irritability, and aberrant motor behavior commonly associated with dementia.16
- A chronic low dose of Δ9-tetrahydrocannabinol (THC) restores cognitive function in old mice.17
- The ECS may protect against aging via homeostatic defense systems such as its antioxidant effects, facilitation of clearance, anti-inflammatory effects, and initiating neuroprotection.18
- The ECS is intricately involved in the process of central and peripheral cells.19
- Beta-Caryophyllene, a cannabis-common terpene and FDA-approved food additive binds with CB2 and protects against cognitive impairment caused by neuro-inflammation. 20
- A high-cholesterol diet reduces the amount of CB1 receptor sites.21
- A growing body of scientific literature implicates the ECS in modulating mitochondrial function22 and, as such, may play a significant role in several chronic pathologies, including the aging process.
- More specifically, CB1 receptors have been located at the membranes of neuronal23 and muscle24 mitochondria controlling cellular respiration and energy production with potential relevance to the aging process.
- Similarly, FAAH, the enzyme that metabolizes AEA, appears to be located on the membrane of the mitochondria and, as such, may play a role in the aging process.25
- CBD may reverse mitochondrial dysfunction by restoring Ca2+ homeostasis.26
- Stress reduction techniques may halt telomere shortening or even extend them. As such, cannabis, famous for its capacity to induce deep relaxation, may significantly mitigate telomere-associated cellular senescence.27
- Anandamide-induced may be a therapeutic strategy in estrogen-deficient conditions (via upregulation of heme oxygenase (HO) and nitric oxide synthase (NOS) enzymes with potential relevance to menopause.28
After this summary about the ECS and general aging, let us look at some of the latest research findings involving sirtuins.
SIRT1– Novel Pathways to Longevity
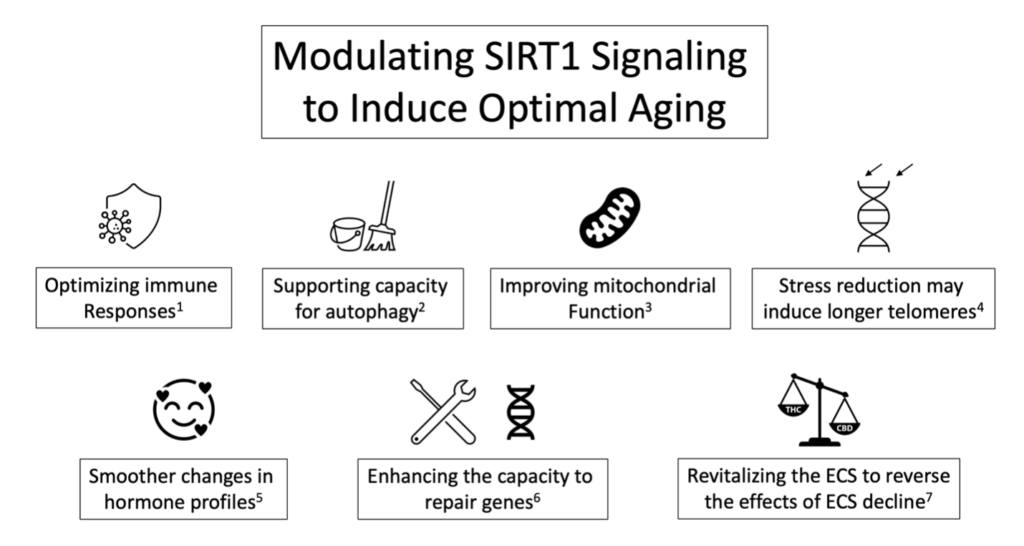
What are Sirtuins, and what role do they play in life extension or anti-aging? Sirtuins are signaling proteins found in all domains of life. However, in humans, a family of seven has been identified (i.e., produced by the genes SIRT 1-7), and to date, SIRT1 has emerged as the one most studied and with the highest potential for life extension.1 Some of the essential effects of SIRT1 in the context of longevity lie in the observations that:
- SIRT1 may prevent age-related diseases and support healthy aging by protecting telomere integrity.2
- Initiate the repair of damaged DNA (via the activation of repair proteins).3
- Generate and maintain cellular integrity and health by inducing optimal epigenetic expressions (i.e., turning on healthy genes and turning off unhealthy ones).4
- Reduces cellular energy stress during aging and age-related diseases.5
- Optimize cellular function.6
- Optimize cellular metabolism relevant to age-related metabolic syndrome, obesity, diabetes, and age-related liver dysfunction (by producing a metabolic shift away from carbohydrates to the use of fat).7
- Mitigate oxidative stress.8
- Detoxification or clearance of old and dying cells (i.e., autophagy) thus supports cellular renewal.9
- Reduces mitochondrial damage and death, and promotes energy metabolism.10
- Similarly to several components of the ECS, SIRT1 expression and activity decrease with sub-optimal aging and in the presence of chronic stressors such as age-related diseases.11
- SIRT1 activation supports optimal immune functions.12
- Homeostatic maintenance and the regulation of lifespan involves SIRT1 via the hypothalamus-pituitary axis.13
The Endocannabinoid System and SIRT1–Research Findings with Potential Clinical Relevance
Up to this point, we explored the roles the ECS and SIRT1 play individually in mitigating the signs and symptoms of pathological aging. Now, we are ready to look at novel research results that offer additional and surprising insights. As it turns out, the reason why modulating components of the ECS with cannabis-based therapeutics (e.g., CBD) work to mitigate sub-optimal aging and, as such, work toward realizing longevity is at least in part due to their ability to support and harness the anti-aging effects initiated by healthy SIRT1 expressions. Consider these preclinical key findings:
- A team of researchers from the Kunming Medical University and Hospital of People’s Liberation Army, Kaiyuan, China (2021) discovered that CBD extends the lifespan and rescues age-associated physiological declines. CBD-induced autophagy protects nerve cells from mitochondrial dysfunction via modulation of SIRT1.1
- A multi-institutional team of Australian scientists, including the Australian Centre for Cannabinoid Clinical and Research Excellence (2022), learned that upregulation in the expression of SIRT1 via the use of CBD resulted in enhanced longevity and neuroprotection.2
- A US-based team from the National Institutes of Health, Bethesda, MD (2020) reported that antagonism at CB1 improves hepatic insulin signaling and glycemic control in obese mice via activation of the SIRT1 (mTORC2/Akt pathway). To provide the reader with some practical context, the cannabis constituent THCV is a known antagonist at CB1.3
- Another group of researchers from the Nanjing Medical University in China (2013) found additional insights implicating CB2. More specifically, agonism at CB2 induced fatty acid oxidation via SIRT1 activation and thus may be a future target in treating conditions that share an underlying pathology of lipid dysregulation.4 To provide the reader with practical context, THC, or the cannabis-common terpene beta-caryophyllene, are known agonists at CB2.
As you can see, CBD is emerging as a significant cannabinoid of interest. Future research will undoubtedly explore any potential effects (e.g., agonism vs. antagonism) cannabinoids (or other cannabis-constituents) can induce via activation of CB1, CB2, other receptor sites or via the modulating of components of the endocannabinoidome (see next paragraph).
SIRT1 and the Endocannabinoidome
Curcumin
Curcumin is a readily available and potent biologically active compound found in turmeric. Curcumin induces SIRT1 upregulation and may benefit age-related conditions such as metabolic syndrome, obesity, and diabetes.1 It is also part of the larger endocannabinoidome, i.e., the physiological environment responsive to changes in the ECS. For instance, while turmeric may not bind with CB1, it affects their actual numbers (expression) and may be quite able to modulate all the effects associated with these receptor sites.2 Emerging research findings suggest a potentially significant value for patients manifesting age-related signs in the form of heart disease3 (e.g., angina, heart attack, reperfusion injuries), Alzheimer’s disease (e.g., neuroprotective, anti-inflammatory, antioxidant, improved mitochondrial functions),4 or the aging of cells that make up the lining of blood vessels and lymphatic vessels.5
Omega-3
The lipidome is a full accounting of all lipids embedded in the membranes of each cell. Lipids are a family of naturally occurring compounds that build much of the internal architecture of living cells. Cellular membranes can be thought of as the skin of the cell. The skin of our trillions of individual cells is made from lipids containing many lipophilic (“fat-loving”) receptor sites such as CB1 or CB2, for example, ready to interact with lipophilic compounds such as cannabinoids. Polyunsaturated fatty acids (PUFAs) are essential building blocks. Since the human body does not make PUFAs, we must consume them to survive and thrive. PUFAs, such as omega-3 and omega-6 fatty acids, are the body’s primary building blocks to make its endocannabinoids. And here is an important insight: a healthy ECS is dependent on the ratio of omega-3 to omega-6. Excess of omega-6 and a lack of dietary omega-3 are implicated in a poorly functioning ECS.6 And, if you need another reason to motivate yourself to eat healthy fat, consider that a high cholesterol diet reduces the amount of available CB1 receptor sites and with it all the potentially beneficial effects associated with them.7
Caloric Restriction
It has been posited that caloric restriction improves longevity and mitigates the development of age-related disorders (e.g., cancer)8 via several mechanisms aimed at the contributing pathologies of aging, including the clearance of accumulating aging and damaged cells (i.e., autophagy), improving mitochondrial energy production, and inducing antioxidant effects, for example. Moreover, everyone can do it inexpensively; all that is needed is the decision and the will to see it through. The good news for those of us who have difficulty sticking to a diet is that using polyphenols produces similar results and is considered a caloric restriction mimetic.9 Polyphenolic compounds (e.g., resveratrol, curcumin, quercetin) contained in dark grapes, turmeric, and fresh capers, for instance, upregulating SIRT1 activity10 and, as such, may produce similar effects as caloric restrictions. In addition, CB1 antagonism (via a synthetic cannabinoid) has been demonstrated to create a synergy alongside caloric restriction and exercise, resulting in weight loss, improved glucose metabolism, and better cardiovascular function11 all-important markers typically used to measure signs of aging.
SIRT1 Cautions
While most of the emerging research results have explored the therapeutic potential of SIRT1, e.g., being a tumor suppressor,1 a few others have produced results that suggest that SIRT1 may act in some cases as or tumor promoter2 suggesting a more complex role (e.g., utilizing either SIRT1 agonists or antagonists) yet to be fully understood.3
In addition, overexpression of SIRT1 may induce potential adverse effects such as worsening inflammation in patients suffering from MS and autoimmune disorders such as rheumatoid arthritis.4
Furthermore, in the context of head and neck cancer, SIRT1 gene silencing decreased ferroptosis (cell death induced by iron-dependent accumulation of lethal lipid peroxidation). In direct contrast, SIRT inducers such as resveratrol increased ferroptosis.5
Summary and Practical Steps to Optimize the Aging Process
While many of the longevity-related trial data is based on preclinical trials, i.e., not conducted on humans, and while much remains to be learned about how specifically cannabinoid-based therapeutics may be utilized to reach for longevity, these early findings and the potential for emerging treatment trends are already pushing the envelope of what we know about aging and the notion of life-extension itself. Here is a summary of related research findings the reader might find instructive:
Cannabidiol (CBD) is a primary cannabinoid that does not produce changes in cognition but tends to provide a gentle uplift in mood. The preclinical trials mentioned above have suggested that CBD-based modulation of various components of the ECS, including that of SIRT1, may produce beneficial effects toward optimal aging.
Delta-9-tetrahydrocannabinol is the primary psychoactive compound found in cannabis. The pre-clinical trials mentioned above have suggested that small amounts of THC may be beneficial, while more significant amounts have produced mixed or detrimental effects. THC activates CB1 and CB2 and, as such, may prevent and mitigates cognitive decline and supports neuroprotection, anti-neuroinflammatory effects, and neurogenesis. The reader is reminded that THC produces the most adverse effects associated with the use of cannabis in a dose-dependent fashion. In other words, the higher the mg dose of THC, the higher the risk of experiencing an adverse effect. THC dosing consideration (based on usage in clinical trials):
- THC microdose: 0.1 mg to 0.4 mg
- THC low dose: 0.5 mg to 5 mg
- THC medium dose: 6 mg to 20 mg
- THC high dose: 21 mg to 50+ mg
While already stated previously but worth repeating here, the endocannabinoidome is the larger internal environment that responds to changes in the ECS that are beyond those initiated by the classical endocannabinoid receptor sites (i.e., CB1 and CB2). These changes may be food-dependent, based on cannabinoid-based therapeutics, or created via mind-body approaches (in part 2). And since their impact can significantly support a healthy aging process, we wanted to include at least those mentioned above at the end of the article.
- Turmeric may increase endocannabinoid receptor site expression and mitigate the age-dependent loss of endocannabinoid receptor sites and signaling.
- Beta-caryophyllene activates CB2 receptor sites and may mitigate are-related neuroinflammation and associated cognitive decline.
- Caloric restrictions, a proven life-extension method, depends, in part, on ECS-based changes in fat cell metabolism.
- Increasing omega-3 intake may make cannabinoids more bioavailable and, as such, may support the beneficial effects associated with them (also consider reducing omega-6 and avoiding unhealthy fats, e.g., trans-fats).
- Reducing stress and increasing activities that induce deep relaxation is significantly modulated via the ECS (in part 2).
In Part 2, we will examine the latest research results focusing on where the cannabinoid health sciences overlap with research results from mind-body medicine and what practical relevance and techniques can be used to mitigate pathological aging and instead harness optimal aging and longevity.
Endnotes: Gerontology–The Science of Aging
1. Bilkei-Gorzo A. (2012). The endocannabinoid system in normal and pathological brain ageing. Philosophical transactions of the Royal Society of London. Series B, Biological sciences, 367(1607), 3326–3341.
Endnotes: The Cannabinoid Health Sciences
1. Devane WA, Dysarz FA 3rd, Johnson MR, Melvin LS, Howlett AC (1988 Nov). Determination and characterization of a cannabinoid receptor in rat brain. Mol Pharmacol. 34(5):605-613.
2. Munro S, Thomas KL, Abu-Shaar M. (1993 Sep). Molecular characterization of a peripheral receptor for cannabinoids. Nature. 365(6441):61-5.
Endnotes: The Endocannabinoid System and Aging in General
Endnotes, graphic (Young/Old Man’s Face):
1. Kataoka K, Bilkei-Gorzo A, Nozaki C, Togo A, Nakamura K, Ohta K, Zimmer A, Asahi T. (2020 Jul) Age-dependent Alteration in Mitochondrial Dynamics and Autophagy in Hippocampal Neuron of Cannabinoid CB1 Receptor-deficient Mice. Brain Res Bull. 160:40-49.
2. Hayakawa K, Mishima K, Nozako M, Hazekawa M, Aoyama Y, Ogata A, Harada K, Fujioka M, Abe K, Egashira N, Iwasaki K, Fujiwara M. (2007 Mar) High-cholesterol feeding aggravates cerebral infarction via decreasing the CB1 receptor. Neurosci Lett. 414(2):183-7.
3. Joaquim HPG, Costa AC, Pereira CAC, Talib LL, Bilt MMV, Loch AA, Gattaz WF. Plasmatic endocannabinoids are decreased in subjects with ultra-high risk of psychosis. Eur J Neurosci. 2021 Oct 29.
Endnotes to text:
1. Bilkei-Gorzo A. (2012). The endocannabinoid system in normal and pathological brain ageing. Philosophical transactions of the Royal Society of London. Series B, Biological sciences, 367(1607), 3326–3341.
Romero J, Berrendero F, Garcia-Gil L, de la Cruz P, Ramos JA, Fernández-Ruiz JJ. (1998 Jun) Loss of cannabinoid receptor binding and messenger RNA levels and cannabinoid agonist-stimulated [35S]guanylyl-5’O-(thio)-triphosphate binding in the basal ganglia of aged rats. Neuroscience. 84(4):1075-83.
Berrendero F, Romero J, García-Gil L, Suarez I, De la Cruz P, Ramos JA, Fernández-Ruiz JJ. (1998 Sep 30) Changes in cannabinoid receptor binding and mRNA levels in several brain regions of aged rats. Biochim Biophys Acta. 1407(3):205-14.
Van Laere K, Goffin K, Casteels C, Dupont P, Mortelmans L, de Hoon J, Bormans G. Gender-dependent increases with healthy aging of the human cerebral cannabinoid-type 1 receptor binding using [(18)F]MK-9470 PET. Neuroimage. 2008 Feb 15;39(4):1533-41.
Wang, L., Liu, J., Harvey-White, J., Zimmer, A., & Kunos, G. (2003). Endocannabinoid signaling via cannabinoid receptor 1 is involved in ethanol preference and its age-dependent decline in mice. Proceedings of the National Academy of Sciences of the United States of America, 100(3), 1393–1398.
2. Long, L. E., Lind, J., Webster, M., & Weickert, C. S. (2012). Developmental trajectory of the endocannabinoid system in human dorsolateral prefrontal cortex. BMC neuroscience, 13, 87.
3. Kataoka K, Bilkei-Gorzo A, Nozaki C, Togo A, Nakamura K, Ohta K, Zimmer A, Asahi T. (2020 Jul) Age-dependent Alteration in Mitochondrial Dynamics and Autophagy in Hippocampal Neuron of Cannabinoid CB1 Receptor-deficient Mice. Brain Res Bull. 160:40-49.
4. Bilkei-Gorzo A, Drews E, Albayram Ö, Piyanova A, Gaffal E, Tueting T, Michel K, Mauer D, Maier W, Zimmer A. Early onset of aging-like changes is restricted to cognitive abilities and skin structure in Cnr1⁻/⁻ mice. Neurobiol Aging. 2012 Jan;33(1):200.e11-22.
5. Carrera, J., Tomberlin, J., Kurtz, J., Karakaya, E., Bostanciklioglu, M., & Albayram, O. (2021). Endocannabinoid Signaling for GABAergic-Microglia (Mis)Communication in the Brain Aging. Frontiers in neuroscience, 14, 606808.
6. Ibid.
7. Ofek, O., Karsak, M., Leclerc, N., Fogel, M., Frenkel, B., Wright, K., Tam, J., Attar-Namdar, M., Kram, V., Shohami, E., Mechoulam, R., Zimmer, A., & Bab, I. (2006). Peripheral cannabinoid receptor, CB2, regulates bone mass. Proceedings of the National Academy of Sciences of the United States of America, 103(3), 696–701.
8. Jiang, W., Zhang, Y., Xiao, L., Van Cleemput, J., Ji, S. P., Bai, G., & Zhang, X. (2005). Cannabinoids promote embryonic and adult hippocampus neurogenesis and produce anxiolytic- and antidepressant-like effects. The Journal of clinical investigation, 115(11), 3104–3116.
9. Goncalves MB, Suetterlin P, Yip P, Molina-Holgado F, Walker DJ, Oudin MJ, Zentar MP, Pollard S, Yáñez-Muñoz RJ, Williams G, Walsh FS, Pangalos MN, Doherty P. (2008 Aug) A diacylglycerol lipase-CB2 cannabinoid pathway regulates adult subventricular zone neurogenesis in an age-dependent manner. Mol Cell Neurosci. 38(4):526-36.
10. Marchalant Y, Brothers HM, Norman GJ, Karelina K, DeVries AC, Wenk GL. Cannabinoids attenuate the effects of aging upon neuroinflammation and neurogenesis. Neurobiol Dis. 2009 May;34(2):300-7.
11. García S, Martín Giménez VM, Mocayar Marón FJ, Reiter RJ, Manucha W. (2020 Aug) Melatonin and cannabinoids: mitochondrial-targeted molecules that may reduce inflammaging in neurodegenerative diseases. Histol Histopathol. 35(8):789-800.
12. Kataoka K, Bilkei-Gorzo A, Nozaki C, Togo A, Nakamura K, Ohta K, Zimmer A, Asahi T. (2020 Jul) Age-dependent Alteration in Mitochondrial Dynamics and Autophagy in Hippocampal Neuron of Cannabinoid CB1 Receptor-deficient Mice. Brain Res Bull. 160:40-49.
13. Pandelides, Z., Thornton, C., Faruque, A. S., Whitehead, A. P., Willett, K. L., & Ashpole, N. M. (2020). Developmental exposure to cannabidiol (CBD) alters longevity and health span of zebrafish (Danio rerio). GeroScience, 42(2), 785–800.
14. Pandelides, Z., Thornton, C., Lovitt, K. G., Faruque, A. S., Whitehead, A. P., Willett, K. L., & Ashpole, N. M. (2020). Developmental exposure to Δ9-tetrahydrocannabinol (THC) causes biphasic effects on longevity, inflammation, and reproduction in aged zebrafish (Danio rerio). GeroScience, 42(3), 923–936.
15. Walther S, Mahlberg R, Eichmann U, Kunz D. (2006 May) Delta-9-tetrahydrocannabinol for nighttime agitation in severe dementia. Psychopharmacology (Berl). 185(4):524-8.
16. Medical Cannabis for the Treatment of Dementia: A Review of Clinical Effectiveness and Guidelines. Ottawa: CADTH; 2019 Jul. (Canadian Agency for Drugs and Technologies in Health rapid response report: summary with critical appraisal).
17. Bilkei-Gorzo, A., Albayram, O., Draffehn, A. et al. (2017) A chronic low dose of Δ9-tetrahydrocannabinol (THC) restores cognitive function in old mice. Nat Med 23, 782–787.
18. Bilkei-Gorzo A. (2012). The endocannabinoid system in normal and pathological brain ageing. Philosophical transactions of the Royal Society of London. Series B, Biological sciences, 367(1607), 3326–3341.
19. Paradisi A, Oddi S, Maccarrone M. (2006 Nov) The endocannabinoid system in ageing: a new target for drug development. Curr Drug Targets. 7(11):1539-52.
20. Kanojia U, Chaturbhuj SG, Sankhe R, Das M, Surubhotla R, Krishnadas N, Gourishetti K, Nayak PG, Kishore A. Beta-Caryophyllene, a CB2R Selective Agonist, Protects Against Cognitive Impairment Caused by Neuro-inflammation and Not in Dementia Due to Ageing Induced by Mitochondrial Dysfunction. CNS Neurol Disord Drug Targets. 2021;20(10):963-974.
21. Hayakawa K, Mishima K, Nozako M, Hazekawa M, Aoyama Y, Ogata A, Harada K, Fujioka M, Abe K, Egashira N, Iwasaki K, Fujiwara M. (2007 Mar) High-cholesterol feeding aggravates cerebral infarction via decreasing the CB1 receptor. Neurosci Lett. 414(2):183-7.
22. Nunn A, Guy G, Bell JD. (2012 Dec 5) Endocannabinoids in neuroendopsychology: multiphasic control of mitochondrial function. Philos Trans R Soc Lond B Biol Sci. 367(1607):3342-52.
Lipina C, Irving AJ, Hundal HS. (2014 Jul) Mitochondria: a possible nexus for the regulation of energy homeostasis by the endocannabinoid system? Am J Physiol Endocrinol Metab. 307(1):E1-13.
Fišar Z, Singh N, Hroudová J. (2014 Nov) Cannabinoid-induced changes in respiration of brain mitochondria. Toxicol Lett. 231(1):62-71.
Singh N, Hroudová J, Fišar Z. (2015 Aug) Cannabinoid-Induced Changes in the Activity of Electron Transport Chain Complexes of Brain Mitochondria. J Mol Neurosci. 56(4):926-931.
Catanzaro G, Rapino C, Oddi S, Maccarrone M. (2009 Oct 16) Anandamide increases swelling and reduces calcium sensitivity of mitochondria. Biochem Biophys Res Commun. 388(2):439-42.
Zhou S, Ling X, Meng P, Liang Y, Shen K, Wu Q, Zhang Y, Chen Q, Chen S, Liu Y, Zhou L. (2021 Sep) Cannabinoid receptor 2 plays a central role in renal tubular mitochondrial dysfunction and kidney ageing. J Cell Mol Med. 25(18):8957-8972.
Zaccagnino P, D’Oria S, Romano LL, Di Venere A, Sardanelli AM, Lorusso M. (2012 Apr) The endocannabinoid 2-arachidonoylglicerol decreases calcium induced cytochrome c release from liver mitochondria. J Bioenerg Biomembr. 44(2):273-80.
Lu Y, Lee DI, Roy Chowdhury S, Lu P, Kamboj A, Anderson CM, Fernyhough P, Anderson HD. (2020 Jan) Activation of Cannabinoid Receptors Attenuates Endothelin-1-Induced Mitochondrial Dysfunction in Rat Ventricular Myocytes. J Cardiovasc Pharmacol. 75(1):54-63.
Oláh A, Alam M, Chéret J, Kis NG, Hegyi Z, Szöllősi AG, Vidali S, Bíró T, Paus R. (. 2020 Jul) Mitochondrial energy metabolism is negatively regulated by cannabinoid receptor 1 in intact human epidermis. Exp Dermatol29(7):616-622.
23. Bénard G, Massa F, Puente N, Lourenço J, Bellocchio L, Soria-Gómez E, Matias I, Delamarre A, Metna-Laurent M, Cannich A, Hebert-Chatelain E, Mulle C, Ortega-Gutiérrez S, Martín-Fontecha M, Klugmann M, Guggenhuber S, Lutz B, Gertsch J, Chaouloff F, López-Rodríguez ML, Grandes P, Rossignol R, Marsicano G. (2012 Mar 4). Mitochondrial CB₁ receptors regulate neuronal energy metabolism. Nat Neurosci. 15(4):558-64.
Hebert-Chatelain E, Desprez T, Serrat R, Bellocchio L, Soria-Gomez E, Busquets-Garcia A, Pagano Zottola AC, Delamarre A, Cannich A, Vincent P, Varilh M, Robin LM, Terral G, García-Fernández MD, Colavita M, Mazier W, Drago F, Puente N, Reguero L, Elezgarai I, Dupuy JW, Cota D, Lopez-Rodriguez ML, Barreda-Gómez G, Massa F, Grandes P, Bénard G, Marsicano G. (2016 Nov 24) A cannabinoid link between mitochondria and memory. Nature. 539(7630):555-559.
Jimenez-Blasco D, Busquets-Garcia A, Hebert-Chatelain E, Serrat R, Vicente-Gutierrez C, Ioannidou C, Gómez-Sotres P, Lopez-Fabuel I, Resch-Beusher M, Resel E, Arnouil D, Saraswat D, Varilh M, Cannich A, Julio-Kalajzic F, Bonilla-Del Río I, Almeida A, Puente N, Achicallende S, Lopez-Rodriguez ML, Jollé C, Déglon N, Pellerin L, Josephine C, Bonvento G, Panatier A, Lutz B, Piazza PV, Guzmán M, Bellocchio L, Bouzier-Sore AK, Grandes P, Bolaños JP, Marsicano G. (2020 Jul) Glucose metabolism links astroglial mitochondria to cannabinoid effects. Nature. 583(7817):603-608.
24. Mendizabal-Zubiaga J, Melser S, Bénard G, Ramos A, Reguero L, Arrabal S, Elezgarai I, Gerrikagoitia I, Suarez J, Rodríguez De Fonseca F, Puente N, Marsicano G, Grandes P. (2016 Oct 25) Cannabinoid CB1 Receptors Are Localized in Striated Muscle Mitochondria and Regulate Mitochondrial Respiration. Front Physiol. 7:476.
25. Morozov YM, Ben-Ari Y, Freund TF. (2004 Jul) The spatial and temporal pattern of fatty acid amide hydrolase expression in rat hippocampus during postnatal development. Eur J Neurosci. 20(2):459-66.
26. Ryan D, Drysdale AJ, Lafourcade C, Pertwee RG, Platt B. (2009 Feb 18) Cannabidiol targets mitochondria to regulate intracellular Ca2+ levels. J Neurosci. 29(7):2053-63.
27. Lengacher CA, Reich RR, Kip KE, Barta M, Ramesar S, Paterson CL, Moscoso MS, Carranza I, Budhrani PH, Kim SJ, Park HY, Jacobsen PB, Schell MJ, Jim HS, Post-White J, Farias JR, Park JY. (2014 Oct) Influence of mindfulness-based stress reduction (MBSR) on telomerase activity in women with breast cancer (BC). Biol Res Nurs. 16(4):438-47.
Schutte NS, Malouff JM, Keng SL. (2020 Aug) Meditation and telomere length: a meta-analysis. Psychol Health. 35(8):901-915.
Innes KE, Selfe TK, Brundage K, Montgomery C, Wen S, Kandati S, Bowles H, Khalsa DS, Huysmans Z. (2018). Effects of Meditation and Music-Listening on Blood Biomarkers of Cellular Aging and Alzheimer’s Disease in Adults with Subjective Cognitive Decline: An Exploratory Randomized Clinical Trial. J Alzheimers Dis. 66(3):947-970.
Dasanayaka NN, Sirisena ND, Samaranayake N. (2021 May). The effects of meditation on length of telomeres in healthy individuals: a systematic review. Syst Rev. 10(1):151.
28. Szabó R, Börzsei D, Szabó Z, Hoffmann A, Zupkó I, Priksz D, Kupai K, Varga C, Pósa A. (2020 Nov) A Potential Involvement of Anandamide in the Modulation of HO/NOS Systems: Women, Menopause, and “Medical Cannabinoids”. Int J Mol Sci. 21(22):8801.
Graphic: Modulating Endocannabinoid Signaling to Induce Optimal Aging
1. Zheng X, Sun T, Wang X. Activation of type 2 cannabinoid receptors (CB2R) promotes fatty acid oxidation through the SIRT1/PGC-1α pathway. Biochem Biophys Res Commun. 2013 Jul 5;436(3):377-81.
2. Kang, S., Li, J., Yao, Z., & Liu, J. (2021). Cannabidiol Induces Autophagy to Protects Neural Cells from Mitochondrial Dysfunction by Upregulating SIRT1 to Inhibits NF-κB and NOTCH Pathways. Frontiers in cellular neuroscience, 15, 654340.
Wang, Z., Zheng, P., Chen, X. et al. Cannabidiol induces autophagy and improves neuronal health associated with SIRT1 mediated longevity. GeroScience (2022).
3. García S, Martín Giménez VM, Mocayar Marón FJ, Reiter RJ, Manucha W. (2020 Aug) Melatonin and cannabinoids: mitochondrial-targeted molecules that may reduce inflammation in neurodegenerative diseases. Histol Histopathol. 35(8):789-800.
Bénard G, Massa F, Puente N, Lourenço J, Bellocchio L, Soria-Gómez E, Matias I, Delamarre A, Metna-Laurent M, Cannich A, Hebert-Chatelain E, Mulle C, Ortega-Gutiérrez S, Martín-Fontecha M, Klugmann M, Guggenhuber S, Lutz B, Gertsch J, Chaouloff F, López-Rodríguez ML, Grandes P, Rossignol R, Marsicano G. (2012 Mar 4). Mitochondrial CB₁ receptors regulate neuronal energy metabolism. Nat Neurosci. 15(4):558-64.
Hebert-Chatelain E, Desprez T, Serrat R, Bellocchio L, Soria-Gomez E, Busquets-Garcia A, Pagano Zottola AC, Delamarre A, Cannich A, Vincent P, Varilh M, Robin LM, Terral G, García-Fernández MD, Colavita M, Mazier W, Drago F, Puente N, Reguero L, Elezgarai I, Dupuy JW, Cota D, Lopez-Rodriguez ML, Barreda-Gómez G, Massa F, Grandes P, Bénard G, Marsicano G. (2016 Nov 24) A cannabinoid link between mitochondria and memory. Nature. 539(7630):555-559.
Jimenez-Blasco D, Busquets-Garcia A, Hebert-Chatelain E, Serrat R, Vicente-Gutierrez C, Ioannidou C, Gómez-Sotres P, Lopez-Fabuel I, Resch-Beusher M, Resel E, Arnouil D, Saraswat D, Varilh M, Cannich A, Julio-Kalajzic F, Bonilla-Del Río I, Almeida A, Puente N, Achicallende S, Lopez-Rodriguez ML, Jollé C, Déglon N, Pellerin L, Josephine C, Bonvento G, Panatier A, Lutz B, Piazza PV, Guzmán M, Bellocchio L, Bouzier-Sore AK, Grandes P, Bolaños JP, Marsicano G. (2020 Jul) Glucose metabolism links astroglial mitochondria to cannabinoid effects. Nature. 583(7817):603-608.
Mendizabal-Zubiaga J, Melser S, Bénard G, Ramos A, Reguero L, Arrabal S, Elezgarai I, Gerrikagoitia I, Suarez J, Rodríguez De Fonseca F, Puente N, Marsicano G, Grandes P. (2016 Oct 25) Cannabinoid CB1 Receptors Are Localized in Striated Muscle Mitochondria and Regulate Mitochondrial Respiration. Front Physiol. 7:476.
4. Lengacher CA, Reich RR, Kip KE, Barta M, Ramesar S, Paterson CL, Moscoso MS, Carranza I, Budhrani PH, Kim SJ, Park HY, Jacobsen PB, Schell MJ, Jim HS, Post-White J, Farias JR, Park JY. (2014 Oct) Influence of mindfulness-based stress reduction (MBSR) on telomerase activity in women with breast cancer (BC). Biol Res Nurs. 16(4):438-47.
Schutte NS, Malouff JM, Keng SL. (2020 Aug) Meditation and telomere length: a meta-analysis. Psychol Health. 35(8):901-915.
Innes KE, Selfe TK, Brundage K, Montgomery C, Wen S, Kandati S, Bowles H, Khalsa DS, Huysmans Z. (2018). Effects of Meditation and Music-Listening on Blood Biomarkers of Cellular Aging and Alzheimer’s Disease in Adults with Subjective Cognitive Decline: An Exploratory Randomized Clinical Trial. J Alzheimers Dis. 66(3):947-970.
Dasanayaka NN, Sirisena ND, Samaranayake N. (2021 May). The effects of meditation on length of telomeres in healthy individuals: a systematic review. Syst Rev. 10(1):151.
5. Szabó R, Börzsei D, Szabó Z, Hoffmann A, Zupkó I, Priksz D, Kupai K, Varga C, Pósa A. (2020 Nov 20) A Potential Involvement of Anandamide in the Modulation of HO/NOS Systems: Women, Menopause, and “Medical Cannabinoids”. Int J Mol Sci. 21(22):8801.
6. Pandelides, Z., Thornton, C., Faruque, A. S., Whitehead, A. P., Willett, K. L., & Ashpole, N. M. (2020). Developmental exposure to cannabidiol (CBD) alters longevity and health span of zebrafish (Danio rerio). GeroScience, 42(2), 785–800.
Kang, S., Li, J., Yao, Z., & Liu, J. (2021). Cannabidiol Induces Autophagy to Protects Neural Cells from Mitochondrial Dysfunction by Upregulating SIRT1 to Inhibits NF-κB and NOTCH Pathways. Frontiers in cellular neuroscience, 15, 654340.
Wang, Z., Zheng, P., Chen, X. et al. Cannabidiol induces autophagy and improves neuronal health associated with SIRT1 mediated longevity. GeroScience (2022).
7. See 1-6 above.
Endnotes: SIRT1– Novel Pathways to Longevity
1. Grabowska, W., Sikora, E., & Bielak-Zmijewska, A. (2017). Sirtuins, a promising target in slowing down the ageing process. Biogerontology, 18(4), 447–476.
2. Osum M, Serakinci N. Impact of circadian disruption on health; SIRT1 and Telomeres. DNA Repair (Amst). 2020 Dec;96:102993.
3. Oberdoerffer, P., Michan, S., McVay, M., Mostoslavsky, R., Vann, J., Park, S. K., Hartlerode, A., Stegmuller, J., Hafner, A., Loerch, P., Wright, S. M., Mills, K. D., Bonni, A., Yankner, B. A., Scully, R., Prolla, T. A., Alt, F. W., & Sinclair, D. A. (2008). SIRT1 redistribution on chromatin promotes genomic stability but alters gene expression during aging. Cell, 135(5), 907–918.
Ming, M., Shea, C. R., Guo, X., Li, X., Soltani, K., Han, W., & He, Y. Y. (2010). Regulation of global genome nucleotide excision repair by SIRT1 through xeroderma pigmentosum C. Proceedings of the National Academy of Sciences of the United States of America, 107(52), 22623–22628.
Wang, R. H., Sengupta, K., Li, C., Kim, H. S., Cao, L., Xiao, C., Kim, S., Xu, X., Zheng, Y., Chilton, B., Jia, R., Zheng, Z. M., Appella, E., Wang, X. W., Ried, T., & Deng, C. X. (2008). Impaired DNA damage response, genome instability, and tumorigenesis in SIRT1 mutant mice. Cancer cell, 14(4), 312–323.
Chung, S., Yao, H., Caito, S., Hwang, J. W., Arunachalam, G., & Rahman, I. (2010). Regulation of SIRT1 in cellular functions: role of polyphenols. Archives of biochemistry and biophysics, 501(1), 79–90.
4. Zhou CH, Zhang MX, Zhou SS, Li H, Gao J, Du L, Yin XX. SIRT1 attenuates neuropathic pain by epigenetic regulation of mGluR1/5 expressions in type 2 diabetic rats. Pain. 2017 Jan;158(1):130-139.
5. Fang, Y., Wang, X., Yang, D., Lu, Y., Wei, G., Yu, W., Liu, X., Zheng, Q., Ying, J., & Hua, F. (2021). Relieving Cellular Energy Stress in Aging, Neurodegenerative, and Metabolic Diseases, SIRT1 as a Therapeutic and Promising Node. Frontiers in aging neuroscience, 13, 738686.
6. Chung, S., Yao, H., Caito, S., Hwang, J. W., Arunachalam, G., & Rahman, I. (2010). Regulation of SIRT1 in cellular functions: role of polyphenols. Archives of biochemistry and biophysics, 501(1), 79–90.
7. Sasaki T. (2015). Age-Associated Weight Gain, Leptin, and SIRT1: A Possible Role for Hypothalamic SIRT1 in the Prevention of Weight Gain and Aging through Modulation of Leptin Sensitivity. Frontiers in endocrinology, 6, 109.
Milne, J. C., Lambert, P. D., Schenk, S., Carney, D. P., Smith, J. J., Gagne, D. J., Jin, L., Boss, O., Perni, R. B., Vu, C. B., Bemis, J. E., Xie, R., Disch, J. S., Ng, P. Y., Nunes, J. J., Lynch, A. V., Yang, H., Galonek, H., Israelian, K., Choy, W., … Westphal, C. H. (2007). Small molecule activators of SIRT1 as therapeutics for the treatment of type 2 diabetes. Nature, 450(7170), 712–716.
Jin, J., Iakova, P., Jiang, Y., Medrano, E. E., & Timchenko, N. A. (2011). The reduction of SIRT1 in livers of old mice leads to impaired body homeostasis and to inhibition of liver proliferation. Hepatology (Baltimore, Md.), 54(3), 989–998.
Li Z, Zhang Z, Ke L, Sun Y, Li W, Feng X, Zhu W, Chen S. (2020 Mar) Resveratrol promotes white adipocytes browning and improves metabolic disorders in Sirt1-dependent manner in mice. FASEB J. 34(3):4527-4539.
Timmers, S., Konings, E., Bilet, L., Houtkooper, R. H., van de Weijer, T., Goossens, G. H., Hoeks, J., van der Krieken, S., Ryu, D., Kersten, S., Moonen-Kornips, E., Hesselink, M., Kunz, I., Schrauwen-Hinderling, V. B., Blaak, E., Auwerx, J., & Schrauwen, P. (2011). Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell metabolism, 14(5), 612–622.
8. Guarente L. Sirtuins in aging and disease. Cold Spring Harb Symp Quant Biol. 2007;72:483-8.
9. Lee, I. H., Cao, L., Mostoslavsky, R., Lombard, D. B., Liu, J., Bruns, N. E., Tsokos, M., Alt, F. W., & Finkel, T. (2008). A role for the NAD-dependent deacetylase Sirt1 in the regulation of autophagy. Proceedings of the National Academy of Sciences of the United States of America, 105(9), 3374–3379.
10. Huang Q, Su H, Qi B, Wang Y, Yan K, Wang X, Li X, Zhao D. A SIRT1 Activator, Ginsenoside Rc, Promotes Energy Metabolism in Cardiomyocytes and Neurons. J Am Chem Soc. 2021 Jan 27;143(3):1416-1427.
11. Fang, Y., Wang, X., Yang, D., Lu, Y., Wei, G., Yu, W., Liu, X., Zheng, Q., Ying, J., & Hua, F. (2021). Relieving Cellular Energy Stress in Aging, Neurodegenerative, and Metabolic Diseases, SIRT1 as a Therapeutic and Promising Node. Frontiers in aging neuroscience, 13, 738686.
12. Hamaidi, I., Kim, S. Sirtuins are crucial regulators of T cell metabolism and functions. (2022) Exp Mol Med 54, 207–215.
13. Yamamoto M, Takahashi Y. (2018 Oct) The Essential Role of SIRT1 in Hypothalamic-Pituitary Axis. Front Endocrinol (Lausanne). 9:605.
Graphic: Modulating SIRT1 Signaling to Induce Optimal Aging
1. Hamaidi, I., Kim, S. Sirtuins are crucial regulators of T cell metabolism and functions. Exp Mol Med 54, 207–215 (2022).
2. Lee, I. H., Cao, L., Mostoslavsky, R., Lombard, D. B., Liu, J., Bruns, N. E., Tsokos, M., Alt, F. W., & Finkel, T. (2008). A role for the NAD-dependent deacetylase Sirt1 in the regulation of autophagy. Proceedings of the National Academy of Sciences of the United States of America, 105(9), 3374–3379.
3. Huang Q, Su H, Qi B, Wang Y, Yan K, Wang X, Li X, Zhao D. A SIRT1 Activator, Ginsenoside Rc, Promotes Energy Metabolism in Cardiomyocytes and Neurons. J Am Chem Soc. 2021 Jan 27;143(3):1416-1427.
4. Osum M, Serakinci N. Impact of circadian disruption on health; SIRT1 and Telomeres. DNA Repair (Amst). 2020 Dec;96:102993.
5. Yamamoto M, Takahashi Y. (2018 Oct) The Essential Role of SIRT1 in Hypothalamic-Pituitary Axis. Front Endocrinol (Lausanne). 9:605.
6. Zhou CH, Zhang MX, Zhou SS, Li H, Gao J, Du L, Yin XX. SIRT1 attenuates neuropathic pain by epigenetic regulation of mGluR1/5 expressions in type 2 diabetic rats. Pain. 2017 Jan;158(1):130-139.
7. Pandelides, Z., Thornton, C., Faruque, A. S., Whitehead, A. P., Willett, K. L., & Ashpole, N. M. (2020). Developmental exposure to cannabidiol (CBD) alters longevity and health span of zebrafish (Danio rerio). GeroScience, 42(2), 785–800.
Kang, S., Li, J., Yao, Z., & Liu, J. (2021). Cannabidiol Induces Autophagy to Protects Neural Cells from Mitochondrial Dysfunction by Upregulating SIRT1 to Inhibits NF-κB and NOTCH Pathways. Frontiers in cellular neuroscience, 15, 654340.
Wang, Z., Zheng, P., Chen, X. et al. Cannabidiol induces autophagy and improves neuronal health associated with SIRT1 mediated longevity. GeroScience (2022).
Bilkei-Gorzo, A., Albayram, O., Draffehn, A. et al. (2017) A chronic low dose of Δ9-tetrahydrocannabinol (THC) restores cognitive function in old mice. Nat Med 23, 782–787.
Pandelides, Z., Thornton, C., Lovitt, K. G., Faruque, A. S., Whitehead, A. P., Willett, K. L., & Ashpole, N. M. (2020). Developmental exposure to Δ9-tetrahydrocannabinol (THC) causes biphasic effects on longevity, inflammation, and reproduction in aged zebrafish (Danio rerio). GeroScience, 42(3), 923–936.
Liu, J., Godlewski, G., Jourdan, T., Liu, Z., Cinar, R., Xiong, K., & Kunos, G. (2019). Cannabinoid-1 Receptor Antagonism Improves Glycemic Control and Increases Energy Expenditure Through Sirtuin-1/Mechanistic Target of Rapamycin Complex 2 and 5’Adenosine Monophosphate-Activated Protein Kinase Signaling. Hepatology (Baltimore, Md.), 69(4), 1535–1548.
Zheng X, Sun T, Wang X. Activation of type 2 cannabinoid receptors (CB2R) promotes fatty acid oxidation through the SIRT1/PGC-1α pathway. Biochem Biophys Res Commun. 2013 Jul 5;436(3):377-81.
Hassanzadeh P, Hassanzadeh A (2012) The CB1 receptor-mediated endocannabinoid signaling and NGF: the novel targets of curcumin. Neurochemical Research 37: 1112–1120.
Zhang Z, Guo Y, Zhang S, et al. Curcumin modulates cannabinoid receptors in liver fibrosis in vivo and inhibits extracellular matrix expression in hepatic stellate cells by suppressing cannabinoid receptor type-1 in vitro. Eur J Pharmacol. 2013;721(1-3):133–140.
Endnotes: The Endocannabinoid System and SIRT1
1. Kang, S., Li, J., Yao, Z., & Liu, J. (2021). Cannabidiol Induces Autophagy to Protects Neural Cells from Mitochondrial Dysfunction by Upregulating SIRT1 to Inhibits NF-κB and NOTCH Pathways. Frontiers in cellular neuroscience, 15, 654340.
2. Wang, Z., Zheng, P., Chen, X. et al. Cannabidiol induces autophagy and improves neuronal health associated with SIRT1 mediated longevity. GeroScience (2022).
3. Liu, J., Godlewski, G., Jourdan, T., Liu, Z., Cinar, R., Xiong, K., & Kunos, G. (2019). Cannabinoid-1 Receptor Antagonism Improves Glycemic Control and Increases Energy Expenditure Through Sirtuin-1/Mechanistic Target of Rapamycin Complex 2 and 5’Adenosine Monophosphate-Activated Protein Kinase Signaling. Hepatology (Baltimore, Md.), 69(4), 1535–1548.
4. Zheng X, Sun T, Wang X. Activation of type 2 cannabinoid receptors (CB2R) promotes fatty acid oxidation through the SIRT1/PGC-1α pathway. Biochem Biophys Res Commun. 2013 Jul 5;436(3):377-81.
Endnotes: SIRT1 and the Endocannabinoidome
1. Zendedel E, Butler AE, Atkin SL, Sahebkar A. Impact of curcumin on sirtuins: A review. J Cell Biochem. 2018 Dec;119(12):10291-10300.
2. Hassanzadeh P, Hassanzadeh A (2012) The CB1 receptor-mediated endocannabinoid signaling and NGF: the novel targets of curcumin. Neurochemical Research 37: 1112–1120.
Zhang Z, Guo Y, Zhang S, et al. Curcumin modulates cannabinoid receptors in liver fibrosis in vivo and inhibits extracellular matrix expression in hepatic stellate cells by suppressing cannabinoid receptor type-1 in vitro. Eur J Pharmacol. 2013;721(1-3):133–140.
3. Yang Y, Duan W, Lin Y, Yi W, Liang Z, Yan J, Wang N, Deng C, Zhang S, Li Y, Chen W, Yu S, Yi D, Jin Z. SIRT1 activation by curcumin pretreatment attenuates mitochondrial oxidative damage induced by myocardial ischemia reperfusion injury. Free Radic Biol Med. 2013 Dec;65:667-679.
4. Sun Q, Jia N, Wang W, Jin H, Xu J, Hu H. (2014 May) Activation of SIRT1 by curcumin blocks the neurotoxicity of amyloid-β25-35 in rat cortical neurons. Biochem Biophys Res Commun. 448(1):89-94.
5. Sun Y, Hu X, Hu G, Xu C, Jiang H. Curcumin Attenuates Hydrogen Peroxide-Induced Premature Senescence via the Activation of SIRT1 in Human Umbilical Vein Endothelial Cells. Biol Pharm Bull. 2015;38(8):1134-41.
6. Freitas HR, Isaac AR, Malcher-Lopes R, Diaz BL, Trevenzoli IH, De Melo Reis RA. Polyunsaturated fatty acids and endocannabinoids in health and disease. Nutr Neurosci. 2018;21(10):695–714.
Calon F, Cole G. Neuroprotective action of omega-3 polyunsaturated fatty acids against neurodegenerative diseases: evidence from animal studies. Prostaglandins LeukotEssent Fatty Acids. 2007;77: 287–293.
Lafourcade M, Larrieu T, Mato S, Duffaud A, Sepers M, Matias I, et al. Nutritionalomega-3 deficiency abolishes endocannabinoid-mediated neuronal functions. NatNeurosci. 2011;14: 345–350.
Larrieu T, Madore C, Joffre C, Layé S. Nutritional n-3 polyunsaturated fatty acids deficiency alters cannabinoid receptor signaling pathway in the brain and associated anxiety-like behavior in mice. J Physiol Biochem. 2012;68: 671–681.
7. Hayakawa K, Mishima K, Nozako M, Hazekawa M, Aoyama Y, Ogata A, Harada K, Fujioka M, Abe K, Egashira N, Iwasaki K, Fujiwara M. (2007 Mar) High-cholesterol feeding aggravates cerebral infarction via decreasing the CB1 receptor. Neurosci Lett. 414(2):183-7.
8. Diaz-Ruiz, A., Di Francesco, A., Carboneau, B. A., Levan, S. R., Pearson, K. J., Price, N. L., Ward, T. M., Bernier, M., de Cabo, R., & Mercken, E. M. (2019). Benefits of Caloric Restriction in Longevity and Chemical-Induced Tumorigenesis Are Transmitted Independent of NQO1. The journals of gerontology. Series A, Biological sciences and medical sciences, 74(2), 155–162.
9. Yessenkyzy, A., Saliev, T., Zhanaliyeva, M., Masoud, A. R., Umbayev, B., Sergazy, S., Krivykh, E., Gulyayev, A., & Nurgozhin, T. (2020). Polyphenols as Caloric-Restriction Mimetics and Autophagy Inducers in Aging Research. Nutrients, 12(5), 1344.
10. Iside, C., Scafuro, M., Nebbioso, A., & Altucci, L. (2020). SIRT1 Activation by Natural Phytochemicals: An Overview. Frontiers in pharmacology, 11, 1225.
11. Lopez Trinidad, L. M., Martinez, R., Kapravelou, G., Galisteo, M., Aranda, P., Porres, J. M., & Lopez-Jurado, M. (2021). Caloric restriction, physical exercise, and CB1 receptor blockade as an efficient combined strategy for bodyweight control and cardiometabolic status improvement in male rats. Scientific reports, 11(1), 4286.
Endnotes: SIRT1 Cautions
1. Powell, M. J., Casimiro, M. C., Cordon-Cardo, C., He, X., Yeow, W. S., Wang, C., McCue, P. A., McBurney, M. W., & Pestell, R. G. (2011). Disruption of a Sirt1-dependent autophagy checkpoint in the prostate results in prostatic intraepithelial neoplasia lesion formation. Cancer research, 71(3), 964–975.
Ming, M., Shea, C. R., Guo, X., Li, X., Soltani, K., Han, W., & He, Y. Y. (2010). Regulation of global genome nucleotide excision repair by SIRT1 through xeroderma pigmentosum C. Proceedings of the National Academy of Sciences of the United States of America, 107(52), 22623–22628.
Kang YY, Sun FL, Zhang Y, Wang Z. SIRT1 acts as a potential tumor suppressor in oral squamous cell carcinoma. J Chin Med Assoc. 2018 May;81(5):416-422.
Leslie M. (2009). SIRT1 takes down tumors. The Journal of Cell Biology, 185(2), 178.
Yeung, F., Hoberg, J. E., Ramsey, C. S., Keller, M. D., Jones, D. R., Frye, R. A., & Mayo, M. W. (2004). Modulation of NF-kappaB-dependent transcription and cell survival by the SIRT1 deacetylase. The EMBO journal, 23(12), 2369–2380.
2. Niederer F, Ospelt C, Brentano F, Hottiger MO, Gay RE, Gay S, Detmar M, Kyburz D. SIRT1 overexpression in the rheumatoid arthritis synovium contributes to proinflammatory cytokine production and apoptosis resistance. Ann Rheum Dis. 2011 Oct;70(10):1866-73.
Lin Z, Yang H, Kong Q, Li J, Lee SM, Gao B, Dong H, Wei J, Song J, Zhang DD, Fang D. USP22 antagonizes p53 transcriptional activation by deubiquitinating Sirt1 to suppress cell apoptosis and is required for mouse embryonic development. Mol Cell. 2012 May 25;46(4):484-94.
Chen WY, Wang DH, Yen RC, Luo J, Gu W, Baylin SB. Tumor suppressor HIC1 directly regulates SIRT1 to modulate p53-dependent DNA-damage responses. Cell. 2005 Nov 4;123(3):437-48.
3. Lin, Z., & Fang, D. (2013). The Roles of SIRT1 in Cancer. Genes & cancer, 4(3-4), 97–104.
4. Niederer F, Ospelt C, Brentano F, Hottiger MO, Gay RE, Gay S, Detmar M, Kyburz D. SIRT1 overexpression in the rheumatoid arthritis synovium contributes to proinflammatory cytokine production and apoptosis resistance. Ann Rheum Dis. 2011 Oct;70(10):1866-73.
5. Lee, J., You, J. H., Kim, M. S., & Roh, J. L. (2020). Epigenetic reprogramming of epithelial-mesenchymal transition promotes ferroptosis of head and neck cancer. Redox biology, 37, 101697.