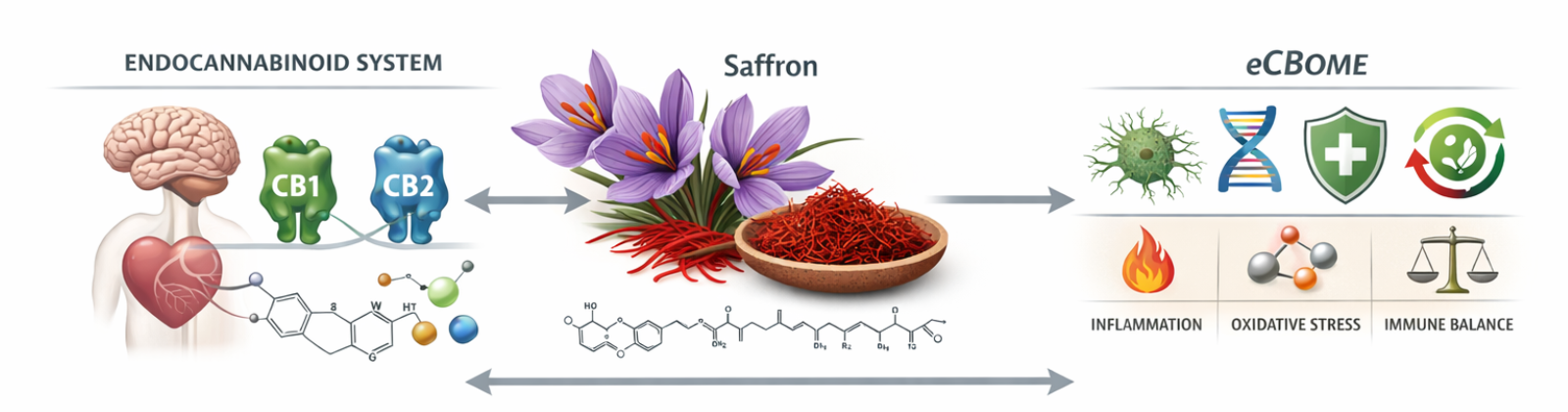
A Regulatory Approach to Inflammation, Mood, and Immune Balance
Most people think of the endocannabinoid system (ECS) as CB1 and CB2 receptors, their endogenous ligands, and the enzymes that synthesize and degrade them. Saffron and the ECS represent a novel and therapeutically relevant area of research, expanding our understanding of immune balance, mood regulation, and inflammatory signaling. Yet biology is rarely confined to isolated pathways.
One helpful way to understand this broader biology is through the concept of the endocannabinoidome (eCBome), the extended network that shapes ECS signaling through interconnected pathways. The eCBome includes metabolic enzymes, immune and inflammatory signaling, redox processes, lipid mediators, gut–brain communication, and neuroendocrine tone. Modulators of the eCBome, whether compounds, behaviors, or environmental factors, can meaningfully influence how endocannabinoid signaling unfolds, often affecting resilience and recovery rather than producing isolated pharmacologic effects.
For example, herbs like turmeric and its main compound curcumin can increase CB2 receptor expression, which helps explain turmeric’s anti-inflammatory effects. Lifestyle changes such as aerobic exercise, yoga, cognitive behavioral therapy (CBT), and healthy eating can also raise levels of anandamide, the endocannabinoid linked to the “runner’s high.”
As we learn more about how to influence the ECS and eCBome, we can start to combine different interventions in smarter ways, especially for chronic conditions that don’t respond well to just one treatment.
Recent studies show that saffron (Crocus sativus), which has a long history in traditional medicine, may uniquely influence both the ECS and the eCBome. Instead of acting like a cannabinoid, saffron seems to shape the environment in which endocannabinoid signaling occurs.
Saffron and the ECS: Beyond CB1 and CB2 Receptors
The main active compounds in saffron—crocin, crocetin, safranal, and picrocrocin—do not attach directly to CB1 or CB2 receptors like typical agonists. Instead, they seem to affect how ECS signaling works during stress, inflammation, and oxidative damage.
When Endocannabinoid Tone Becomes Dysregulated
Normally, the ECS helps protect the body. But if ECS signaling becomes suboptimal, it can lose accuracy, leading to ongoing inflammation or a slow immune recovery. This occurs in conditions characterized by chronic inflammation and oxidative stress, including neurodegenerative, metabolic, autoimmune, and stress-related disorders. Emerging evidence suggests that viral and immune-mediated conditions are also associated with altered endocannabinoid signaling dynamics. While these changes might help control inflammation at first, if they last too long, they can get in the way of healing and cause tissue damage.
This risk may be greater with high-potency full agonists, particularly certain synthetic cannabinoids, which can override endogenous feedback regulation.
Oxidative Stress and Endocannabinoid Disruption
Oxidative stress is a main factor in this process. Immune cells like macrophages use reactive oxygen species (ROS), including hydrogen peroxide (H₂O₂), to kill pathogens. Normally, this burst of oxidation is carefully controlled and quickly neutralized by antioxidant systems such as superoxide dismutase, catalase, and glutathione peroxidase, which protect healthy tissue. Most pathogens lack these defenses, making them more vulnerable to oxidative damage.
However, if oxidative stress is not resolved, excess free radicals can damage cell membranes, proteins, DNA, and mitochondria, which are not designed to withstand sustained oxidative attack. In modern environments marked by chronic stress, metabolic dysfunction, and environmental exposures, long-term oxidative stress is increasingly common, further disrupting ECS signaling and amplifying chronic inflammatory states.
Saffron as a Modulator of Endocannabinoid Tone and Neuroinflammation
In a controlled preclinical study of retinal neurodegeneration, light injury caused abnormal increases in CB1 and CB2 receptor expression, a pattern also reported in various chronic inflammatory and immune-mediated conditions. Saffron treatment brought CB1 and CB2 gene and protein expression back to normal, reduced photoreceptor cell death, and protected the retina’s structure and function. This study was one of the first to directly connect saffron’s neuroprotective effects to changes in the ECS and the wider eCBome (R. Maccarone et al., 2016).
Simply put, saffron did not “activate” the ECS. Instead, it helped bring the system back into balance.
This shows a key idea of the eCBome: it is better to support the body’s natural balance than to push just one signaling pathway.
CB2 Signaling, Neuroinflammation, and Immune Regulation
The same study found that CB2 played a bigger role than CB1 in providing neuroprotection, which matches what we know about CB2’s part in controlling microglia, chemokine signals, and immune recovery (R. Maccarone et al., 2016).
Saffron treatment greatly lowered the expression of CCL2, a chemokine that acts as a distress signal to bring immune cells to injured tissue. This response helps at first, but if CCL2 stays high, it can cause long-term inflammation and extra tissue damage. Interestingly, blocking CB1 and CB2 also lowers CCL2, which suggests that saffron and ECS-targeted treatments may work through some of the same inflammation control pathways (R. Maccarone et al., 2016).
In short, saffron appears to dampen pro-inflammatory signaling, helping the immune system settle rather than overreact.
This places saffron at the intersection of:
- ECS signaling
- Neuroinflammation (saffron and crocin suppress inflammatory mediators)
- Oxidative stress (crocin, crocetin, and safranal amplify antioxidant capacity)
- Immune regulation
All of these areas are part of what the eCBome does.
Shared Downstream Pathways: PPAR-γ, NF-κB, SIRT1, and Nrf2 Signaling
Beyond direct receptor interactions, saffron constituents and cannabinoids such as THC, CBD, CBG, and the endocannabinoid-like lipid PEA converge on several shared downstream regulatory pathways. These include:
- Crocin restored peroxisome proliferator-activated receptor gamma (PPAR-γ) expression (a nuclear receptor that regulates metabolism, inflammation, and cellular differentiation) that had been suppressed under diabetic inflammatory conditions. Crocin also reduced tumor necrosis factor alpha (TNF-α) (a pro-inflammatory cytokine) and interleukin-6 (IL-6) levels (pro-inflammatory immune signaling molecules), improved antioxidant capacity, and lowered cardiac injury markers. The reversal of these effects by the PPAR-γ antagonist GW9662 suggests that crocin’s protective actions are mediated, at least in part, through PPAR-γ activation (M. Badavi et al., 2020).
Inflammatory and Immune Signaling Pathways
- Crocin, crocetin, and safranal exert immunoregulatory effects by antagonizing nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) (a central pro-inflammatory transcription factor), reducing TNF-α, interferon gamma (IFN-γ) (a cytokine involved in immune activation and antiviral defense), and interleukins such as IL-1β, IL-6, IL-12, and IL-17A (key mediators of inflammatory signaling). Saffron has also been described as an agonist of peroxisome proliferator-activated receptor gamma (PPAR-γ) (a nuclear receptor that regulates metabolism, immune balance, and inflammatory resolution), while down-regulating pro-inflammatory enzymes including inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2). Through these coordinated mechanisms, saffron constituents may modulate key mechanisms involved in the pathogenesis of immune-mediated disorders by restoring immune and inflammatory balance and supporting modulation of both cellular and humoral immune responses (M. Zeinali et al., 2019).
Redox and Cellular Defense Signaling
- Saffron and crocin improve inflammatory reactions and oxidative stress by up‐regulation of SIRT1 (a longevity-associated enzyme that regulates cellular stress response, inflammation, and mitochondrial function) and Nrf2 signaling (a transcription factor that activates antioxidant and cellular defense genes) (F. Anaeigoudari et al., 2023)
- Crocin inhibits inducible nitric oxide synthase (iNOS) (an enzyme that produces nitric oxide during inflammation and immune activation) and cyclooxygenase-2 (COX-2) (an inducible enzyme that drives production of pro-inflammatory prostaglandins) and thus induces anti‐inflammatory and antioxidant effects (F. Anaeigoudari et al., 2023)
- Saffron and several of its constituents modulate calcium (Ca2+) and potassium (K⁺) channels, resulting in smooth muscle relaxation and vasodilation with potential use in patients challenged by high blood pressure (N. Al-Saigh et al., 2022; M. Boskabady et al., 2020)
- Safranal binds with serotonin receptor sites 5-HT1A and 5-HT2A, resulting in antidepressant and anxiolytic effects (F. Amin et al., 2022)
- Crocin modulates gamma-aminobutyric acid (GABA-A) receptors, resulting in anxiolytic and anti-depressant effects (R. Matraszek-Gawron et al., 2022; T. E. Ceremuga et al., 2018)
These shared pathways help explain why saffron lowers oxidative stress and inflammation, making it easier to optimize endocannabinoid signaling.
Why This Matters for Endocannabinoid Medicine
Clinical Relevance: Oxidative Stress, Mood, and Immune Dysregulation
Clinically, this regulatory profile suggests saffron may be particularly relevant in contexts characterized by oxidative stress, immune dysregulation, and suboptimal endocannabinoid tone, including mood disorders, metabolic dysfunction, neuroinflammation, and stress-related syndromes. Rather than targeting a single symptom, saffron appears to influence the broader regulatory environment in which symptoms arise.
Saffron as a Natural eCBome Modulator in Endocannabinoid Medicine
In endocannabinoid medicine, saffron works less like a drug and more like a modulator that shapes the body’s internal environment. Research suggests it may:
- Limit ECS overactivation in disease states
- Support CB2-mediated immune resolution
- Promote neuroprotection without receptor overstimulation
This makes saffron a good choice to use together with cannabinoids like CBD or THC, ECS-related compounds like PEA, endocannabinoid precursors such as omega-3 fatty acids, and lifestyle changes.
In regulatory medicine, synergy is not about stacking effects, but about restoring signal fidelity. By lowering oxidative noise and inflammatory distortion, saffron may allow endogenous and plant-derived cannabinoids to operate within a more physiologically coherent environment.
Rather than forcing receptor activation, this approach supports tone normalization, immune resolution, and redox balance — conditions that allow the endocannabinoid system to resume its adaptive, self-regulating role. In this way, saffron supports a return to physiological balance rather than merely mitigating one specific symptom of complex diseases.
Clinical Implications for Endocannabinoid Medicine
Research on saffron and the ECS highlights a broader principle: restoration of physiological balance often matters more than direct receptor activation. The ECS does not operate in isolation; it is part of the larger eCBome, where plant compounds, redox balance, immune signaling, and emotional regulation interact to shape overall health.
Sometimes, the most effective intervention is not to amplify a signal, but to restore the conditions under which balance can re-emerge on its own.
Saffron may be one of those quiet allies.
Endnotes:
Amin F, Ibrahim MAA, Rizwan-Ul-Hasan S, Khaliq S, Gabr GA, Muhammad, Khan A, Sidhom PA, Tikmani P, Shawky AM, Ahmad S, Abidi SH. Interactions of Apigenin and Safranal with the 5HT1A and 5HT2A Receptors and Behavioral Effects in Depression and Anxiety: A Molecular Docking, Lipid-Mediated Molecular Dynamics, and In Vivo Analysis. Molecules. 2022 Dec 7;27(24):8658. doi: 10.3390/molecules27248658. PMID: 36557792; PMCID: PMC9783496.
Anaeigoudari F, Anaeigoudari A, Kheirkhah-Vakilabad A. A review of therapeutic impacts of saffron (Crocus sativus L.) and its constituents. Physiol Rep. 2023 Aug;11(15):e15785. doi: 10.14814/phy2.15785. PMID: 37537722; PMCID: PMC10400758.
Badavi M, Mard SA, Dianat M, Dashtbozorgi N. Crocin attenuates oxidative stress and inflammation in myocardial infarction induced by isoprenaline via PPARγ activation in diabetic rats. J Diabetes Metab Disord. 2020 Nov 18;19(2):1517-1525. doi: 10.1007/s40200-020-00686-y. PMID: 33553037; PMCID: PMC7843838.
M. Boskabady et al., Chapter 17 – Crocus sativus L. (Saffron) and Its Components Relaxant Effect on Smooth Muscles and Clinical Applications of This Effect, Academic Press, 2020, Pages 219-231, ISBN 9780128184622.
Ceremuga TE, Ayala MP, Chicoine CRW, Chun CSM, DeGroot CJM, Henson DT, Randall CSA, Stanley CLR, Beaumont CDM. Investigation of the Anxiolytic and Antidepressant Effects of Crocin, a Compound from Saffron (Crocus sativus L), in the Male Sprague-Dawley Rat. AANA J. 2018 Jun;86(3):225-233. PMID: 31580812.
Maccarone R, Rapino C, Zerti D, di Tommaso M, Battista N, Di Marco S, Bisti S, Maccarrone M. Modulation of Type-1 and Type-2 Cannabinoid Receptors by Saffron in a Rat Model of Retinal Neurodegeneration. PLoS One. 2016 Nov 18;11(11):e0166827.
Zeinali M, Zirak MR, Rezaee SA, Karimi G, Hosseinzadeh H. Immunoregulatory and anti-inflammatory properties of Crocus sativus (Saffron) and its main active constituents: A review. Iranian Journal of Basic Medical Sciences. 2019 Apr;22(4):334-344. DOI: 10.22038/ijbms.2019.34365.8158. PMID: 31223464; PMCID: PMC6535192.